A Unique Out-licensing Opportunity: A Novel Pancreatic Cancer Early Diagnostic Technology
Author: Patrick Page, Corporate Development Director
JSR Life Sciences (JLS), the parent company of Crown Bioscience, provides specialized technology, products, materials, and services to the biopharmaceutical industry and academia-based life science researchers.
JLS scientists recently developed a unique in vitro pancreatic cancer early detection test (iv-PCED) that has great potential for positioning as a direct-to-consumer (DtC) cancer genetic test or medical cancer genetic test (see Figure 1). JLS is seeking interested parties to out-license this unique asset to a partner with expertise and experience in developing genetic cancer diagnostic tests so it can continue its development for eventual marketing.

Figure 1: Potential Positioning of the iv-PCED as a Direct-to-Consumer and/or Medical Cancer Genetic Test.
Capturing Diagnostic (Dx) Value in Early Cancer Detection
The novel iv-PCED can capture value in the growing field of early cancer detection, which has witnessed an increasing number of diagnostic (Dx) tests under development or commercially available. Since survival greatly improves when cancer is detected early, and therapies are becoming increasingly expensive, there is a strong rationale for payors to reimburse for such technologies and ensure proper use of therapeutics. Furthermore, early cancer detection can provide healthcare providers with valuable insights that can guide optimal care for their patients, who are generally becoming better informed, empowered, and tech-savvy and want access to better Dx testing options. Also, the Dx landscape for early cancer detection is evolving rapidly and presents exciting growth opportunities, especially for genetic cancer diagnostic tests based on powerful next generation sequencing (NGS) technology.
NGS as a Genetic Cancer Dx Test
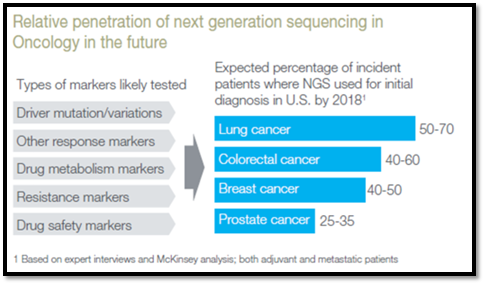
Figure 2: Relative Penetration of NGS Sequencing in Oncology.
Estimates indicate that the adoption of NGS for clinical Dx is 25–70% of newly diagnosed cancer patients (depending on tumor type) (Figure 2). For highly heterogenous diseases, such as cancer, NGS-based technologies can play a role not only in early detection and diagnosis, but also for prognosis and to monitor changes in genetic profiles during disease progression and treatment.
Furthermore, interest and opportunity are increasing for liquid biopsies which are blood-based tests that can detect cancer cells or DNA circulating in the blood and can reduce reliance on highly invasive surgical biopsies. Since early detection of cancer is closely tied to better outcomes and survival, it is no surprise that several NGS-based multigene panels have been developed commercially. As shown below, NGS is relevant to many areas of oncology, including a variety of types of genetic markers that are applicable to multiple tumor types.
Early Diagnosis of Pancreatic Cancer Remains an Unmet Need
Pancreatic cancer incidence is rising by about 1% per year, and it accounts for ~3% of all cancers and 7% of cancer mortality in the United States. Pancreatic cancer is now the third leading cause of cancer-related death, and it is exceptionally lethal in part because it is challenging to detect at an early stage. By the time signs and symptoms appear, the disease has often already reached an advanced stage and metastasized. The 5-year survival rate is dismal at 12%, and no clinically relevant pancreatic cancer screening test can reliably detect early-stage cancer in individuals without symptoms.
Novel in vitro Pancreatic Cancer Early Detection Test (iv-PCED) as a Disruptive Dx Asset
JLS scientists have identified and validated a panel of biomarkers that are relevant for the early diagnosis of pancreatic cancer. This novel approach, protected by intellectual property, uses a unique genetic cancer biomarker panel consisting of 25 specific fusion points shown to be relevant, in combination with an integrated and proprietary artificial intelligence (AI) algorithm to analyze the fusion points and provide valuable insights. This biomarker panel and companion AI holds great commercial potential as a Dx tool, such as for an in vitro PCED test (Figure 3).
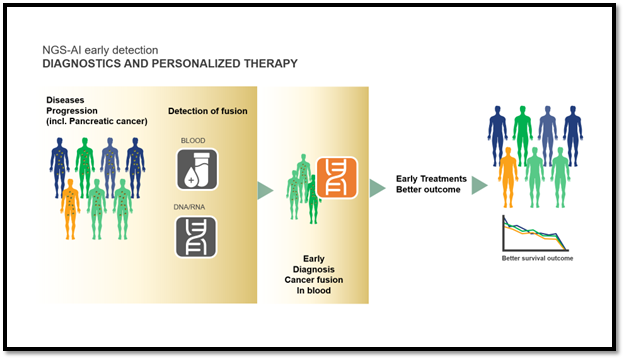
Figure 3: Potential Use of the iv-PCED.
Based on the preliminary investigation, JLS in vitro PCED test will have high selectivity and specificity and may be positioned as a DtC Dx targeting asymptomatic, high-risk individuals (such as those older than 50) and/or a medical Dx test for younger individuals with a family history of pancreatic cancer. Conservative estimates indicate a 250+ million to multibillion market per year. Differential pricing and reimbursement figures are expected for a positioning as DtC iv-PCED test or medical Dx test. The table below highlights the target product profile of the asset (Table 1).
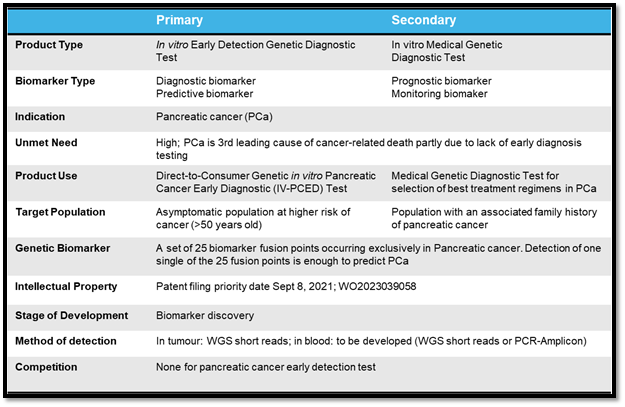
Table 1: Target Product Profile of the iv-PCED.
A Variety of Potential Collaborative Out-licensing Models
JLS understands the challenges associated with genetic-based Dx test development and is open to negotiating win-win licensing terms and conditions with potential acquirers of this technology. JLS could provide a standard license or collaborative licensing model, offering the licensee a creditable note against up-front and milestone payments on expenses incurred with JLS affiliated companies, such as Crown Bioscience and MBL, that can provide support and services to potential buyers as they develop the Dx test.
To learn more about this unique out-licensing opportunity, please contact outlicensing@crownbio.com